Cologuard is a screening test for colon and rectal cancer. The cancer cells may have DNA changes in.
 Colorectal Cancer Screening Science Should Trump Convenience Stat
Colorectal Cancer Screening Science Should Trump Convenience Stat
Detection of colorectal neoplasia associated DNA markers and for the presence of occult haemoglobin in human stool.
Cologuard stool test. In August 2014 the US. These cells pass with the stool through the colon. This recently FDA approved test is becoming available in the majority of US states and territories.
Colon cancer screening guideline recommendations vary for people under the age of 50 and over the age of 75. The colon sheds cells from its lining every day. Cologuard is intended for the qualitative detection of colorectal neoplasia associated DNA markers and for the presence of occult hemoglobin in human stool.
The three most common tests are an annual fecal immunochemical test or fecal occult blood test FITFOBT that detects blood in the stool. Food and Drug Administration FDA approved the companys at-home multitarget stool DNA screening test called Cologuard for the detection of colorectal cancers and pre-cancers. Cologuard looks for changes in your DNA that could indicate the.
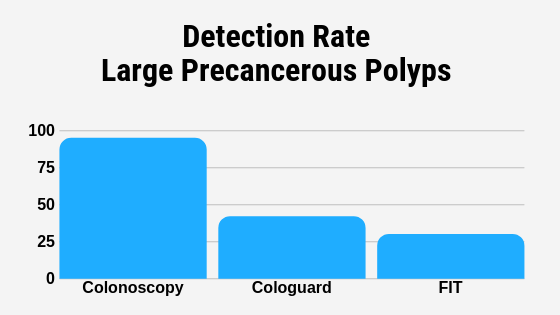
For the first time the Centers for Medicare Medicaid Services CMS simultaneously proposed national coverage for the test as part of a pilot FDA-CMS parallel review program. It is noninvasive meaning it is done without putting any screening tool into the body and looks at the DNA in your stool to determine if cancer or precancerous cells exist. Cologuard also checks for precancerous polyps that might be present in your colon.
A stool test is one of many tests that may be used to screen for colon cancer. Cologuard is the recently Food and Drug Administration FDA-approved stool deoxyribonucleic acid DNA screening test for detecting colon cancer. Talk to your doctor about what puts you.
The presence of abnormal cells or blood in the stool may indicate cancer or precancer tumors. The colon sheds cells from its lining every day. Cologuard is a screening test for colon and rectal cancer.
The Cologuard test is a stool DNA test that looks for microscopic blood in the stool and altered DNA. Cologuard is the only stool-DNA screening test for detecting colon cancer that is approved by the Food and Drug Administration FDA. The test looks for changes in your DNA that could indicate the presence of colon cancer.
Its performed using an at-home kit that includes a container for a stool sample. The multitarget stool DNA mt-sDNA test Cologuard. Learn more about Cologuard now.
Cologuard detects the altered DNA. Cologuard is a screening test for colon and rectal cancer. The colon sheds cells from its lining every day.
The American Cancer Society ACS includes multi-target stool DNA Cologuard as one of the CRC screening options for average risk adults ages 45 and older. Cologuard is the only stool-DNA screening test for detecting colon cancer that is approved by the Food and Drug Administration FDA. That sample is then shipped to a lab for testing.
These cells pass with the stool through the colon. The cancer cells may have DNA changes in certain genes. The Colorguard test is different from a colonoscopy because you can do it at home and you do not need to prepare by fasting or discontinuing medications before or after the test.
The cancer cells may have DNA changes in. These cells pass with the stool through the colon. Cologuard is a new colon cancer screening method.
Cologuard is a noninvasive colon cancer screening test for people 45 years of age and older who are at average risk for colon cancer. Which screening test you choose depends on your risk your preference and your doctor. In August 2014 the Food and Drug Administration FDA approved Cologuard a noninvasive multitarget stool DNA screening test for colorectal cancer CRC.
ACS recommends Cologuard every 3 years. Other tests include sigmoidoscopy colonoscopy and CT colonography. A positive result may indicate the presence of colorectal cancer CRC or advanced adenoma AA and should be followed by diagnostic colonoscopy.


